A novel redox-based mechanism that controls Tau aggregation has been identified by the Papanikolopoulou lab, providing new insights into how Tau can adopt a highly aggregated conformation in vivo.
A new study published in Acta Neuroapathologica Communications by the Papanikolopoulou lab revealed the role of the in vivo post-translational modification of Tau’s cysteines residues by the Mical enzyme in Tau function and dysfunction.
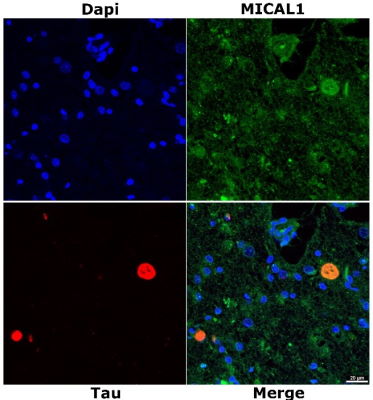
Tau accumulation is clearly linked to Tau pathogenesis in Alzheimer’s disease and other Tauopathies. However, processes leading to Tau fibrillization and reasons for its pathogenicity remain largely elusive. In a new study published in Acta Neuroapathologica Communications entitled “Mical regulates Tau toxicity via cysteine oxidation in vivo”, Papanikolopoulou and colleagues use genetic interactions and mass spectrometry analysis to show that Mical oxidizes Tau at Cys322 and greatly affects its aggregation propensity and Tau-associated neuronal toxicity and dysfunction. Importantly, they validate their results from the fly model in human Tauopathy samples and show that MICAL1 is up-regulated in the brains of Tauopathy patients and co-localizes with Tau in Pick bodies. Tau oxidation by Mical likely defines a critical event in Tau pathogenesis and inhibition of Mical would in principle offer a new intervention approach.
Engie Prifti , Eleni N Tsakiri, Ergina Vourkou, George Stamatakis, Martina Samiotaki, Efthimios M C Skoulakis, Katerina Papanikolopoulou. Mical modulates Tau toxicity via cysteine oxidation in vivo. Acta Neuropathol Commun (2022) 10(1):44. doi: 10.1186/s40478-022-01348-1.